Translational Awards aim to progress the development of novel, innovative technologies towards benefits to human cardiovascular health. The scheme supports the development of technologies with transformative potential from the proof-of-concept stage to being ready for the commercial market.
Proposals are expected to demonstrate a strong intellectual property position and a development proposition that could attract follow-on investment.
Projects can start and finish at any point along the translational pathway from initial development, through pre-clinical studies to early phase clinical trials (up to and including phase II). The finishing point must represent a significant scientific and/or value inflection point.
Please ensure you have read the information on this page before you start an application on the BHF Grant Management System.
Use the links below to jump straight to the information you require.
Who can apply
- The principal investigator will be a researcher working in an established research institution in the UK.
- Individuals or teams from commercial organisations can apply as collaborators.
Budget and grant duration
- There is no limit on the amount of funding that can be applied for, but it should be justified and appropriate to the project. Applicants are strongly encouraged to contact BHF Translational Awards Team to discuss potential application.
- Up to 3 years, with clearly defined progression milestones (typically 3 - 5 milestones). Applicants must justify the timescale in the context of the proposed work. Applicants should note that continued funding will be subject to the achievement of the relevant milestones.
What we are looking for
- The project should be novel and inventive and address an unmet clinical need within the BHF remit.
- The project should result in a product, technology or intervention that is ready for market development.
- The project should be milestone-driven and have a credible delivery plan.
We do not expect milestones to overlap. However, in
exceptional circumstances we may be prepared to consider overlapping milestones
if they can be appropriately justified.
The scheme covers any type of technology, including:
- novel therapeutics (small molecules or biologics)
- drug repurposing
- devices
- surgical techniques
- diagnostics
- imaging technologies
- digital solutions
- cell therapy and regenerative medicine
- early-phase clinical studies evaluating efficacy and safety.
The scheme does not support funding for investigative research focussed on open-ended questions and late-phase clinical trials. For those purposes, refer to either Project Grants or Clinical Studies funding schemes.
The application should provide clear and compelling information on the following:
- an unmet healthcare need in heart and circulatory disease
- the advantages of the proposed technology over competing solutions
- supporting proof of concept data
- a structured project with non-overlapping milestones, each with SMART GO/NO-GO success criteria, and an estimate of costs per milestone. We do not expect milestones to overlap. However, in exceptional circumstances we may be prepared to consider overlapping milestones if they can be appropriately justified. Milestone success criteria should be SMART (specific, measurable, achievable, relevant, timely), and detail any robust GO/NO-GO criteria (failure to meet these criteria will result in early termination of the project). For all projects, it is advisable to structure the project so that the critical question(s) are addressed as early as possible in the plan.
- the intellectual property position
- a proposed route to market and commercialisation
- a strategy to navigate the relevant regulatory processes
- long term aims beyond the BHF Translational Award.
We require that you work with your Institution's Technology Transfer Office (TTO) to develop the intellectual property strategy and commercial case for the application. It is important to consider the entire route to commercialisation for your technology and not just what may be funded by the particular Translational Award requested. Providing this information gives the BHF and subsequent investors the confidence that the project is well designed and has the best chance of success.
The MHRA Innovation Office provides a free, informal service that can help with your regulatory queries.
Are you unsure whether your research is at the appropriate stage of development or is eligible for funding? Please contact a member of the Research Translation Team who will be able to give specific advice regarding the project.
Back to top ^
What we will fund
The award can be used flexibly. We will consider funding staff salaries (excluding PhD studentships), consumables and essential research equipment, consultancy fees, outsourcing to contract research organisations and other fee-for service providers.
Outsourcing
We will consider providing funds to outsource project work to parties who undertake work on a contracted/out-sourced basis (e.g. contract research organisations (CROs)), subject to appropriate justification. Such parties may carry out specifically defined piece(s) of work on behalf of the investigators on a fee-for-service basis, with no potential claim as an inventor over any arising intellectual property.
Project management
Successful Translational Awards require efficient management and reporting to ensure that the project stays on target. An experienced project manager (salary grade 7 or 8) may be appointed for up to 0.2FTE per annum to assist with this. They will be responsible for managing and coordinating project activities and communications within your organisation and between collaborators and sub-contractors, as well as regularly reporting to the BHF.
Please find here more information on costing a grant.
Intellectual property management
Where possible, intellectual property generated in the course of BHF Translational Award project will be owned by the host institute, who will have the right to manage and exploit the project’s intellectual property. We expect you to have a strategy on how to protect, maintain and commercialise BHF-funded intellectual property and provide any costs for such activity.
Back to top ^
How to apply
Applications are assessed in a two-stage process and must be completed in partnership with your Institution's TTO.
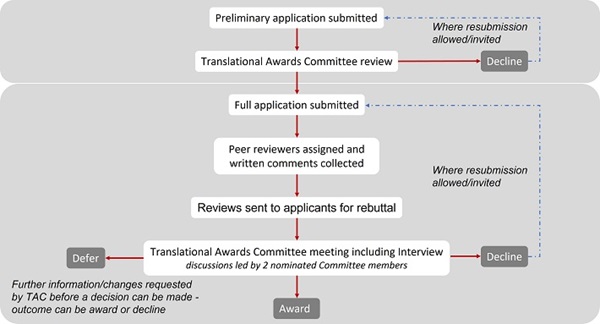
1. Submit your preliminary application
Applicants are required to submit a preliminary application which will be considered by the Translational Awards Committee. To access the Translational Award preliminary application form, please log in to the BHF Grants Management System.
For further guidance, refer to the Translational Award Preliminary Application Sample. Following a successful preliminary application, the applicant will be invited to submit a full application form.
2. Submit your full application
Full applications will be assessed by external peer reviewers and considered by the Translational Awards Committee. In addition, applicants will be invited to present and defend their research proposal at interview by the Translational Awards Committee.
Refer to The Translational Award Full Application Sample
Back to top ^
How we will assess your application
Preliminary applications are reviewed by members of the Translational Awards Commitee.
Full applications are reviewed by independent expert reviewers and the Translational Awards Commitee.
Please allow up to 10 months from the preliminary application submission to final funding decision.
Independent expert review
To help applicants better understand how their proposals are assessed, please see BHF independent review guidelines and blank review form.
The flowchart below shows the application journey.
Application submission deadlines
There are two opportunities to submit preliminary or full applications each year:
Applications must be received by 17:00 on the deadline date. Late submissions will not be considered.
We will acknowledge receipt of your application within 48 hours and advise when you can expect the result.
BHF reserve the right to change the deadline dates. We recommend that you check this page on a regular basis to take note of any changes.
Apply online
Back to top ^
Useful links
BHF have partnered with Panacea Innovation to launch CardioStars, a unique programme that will equip researchers with the entrepreneurial and business skills to become the science innovators of tomorrow.
Find out how to apply for a research grant from the British Heart Foundation. Check our grant types and read through the relevant grant guidelines
Clinical Trials Units (CTUs) are specialist units that help clinical researchers design, conduct, analyse and publish clinical trials.
This page explains our policies on registering clinical trials on a database, how to manage a clinical trial and what the BHF expects with regard to data sharing for clinical trials.
Back to top ^