Clinical Research Training Fellowships
Entry requirements
- Applicants should usually be after Foundation year two, but before award of a CCT.
- Residency requirements do not apply
- Clinical Research Training Fellowship candidates are eligible to apply for accelerated endorsement of the UK Global Talent Visa. More information on the UK Global Talent Visa
Grant duration
- Usually three years to complete a PhD, although applications for two years to complete an MD or equivalent will be considered.
Award may include
- Salary of applicant
- Research consumables directly attributable to the project, usually less than £18,000 per year
- Tuition fees (excluding college fees) at standard home student rate (set by the research institution, although a cap applies. Please refer to the Grant Costing Guide).
- Travel funding of up to £3,000 per award to present research at or attend scientific meetings relevant to the grant
How to apply
- Read the information in How to apply
- Read our commitment to Improving Support for Clinical Academics
- If you are applying for funding for a clinical study, also read:
- Log onto the online application form
- Complete all the sections of the online application form
Who can apply?
- The application must be made by an established investigator who will be the supervisor for a named student. The primary supervisor must devote a minimum of 10% of their time to supervising the student and hold a post at the host institution for the duration of the studentship. A second supervisor should also be included.
- Applicants who have started a PhD (or equivalent) may be eligible to apply for a Clinical Research Training Fellowship, but may only apply within 6 months of having registered for their PhD. In these circumstances, the proposed start date for the Clinical Research Training Fellowship should also be no more than 12 months (or full-time equivalent) from the date of registration for the degree.
Case for Support
You will be asked to attach a single PDF document to your online application form containing the following information:
- For resubmissions, include an unedited copy of the original feedback followed by a detailed response, limited to 3 sides of A4, explaining how the revised application has changed from the original submission.
- Title of the proposed research
- Abstract of the proposed investigation in 200 words or less
- Background to the project and pilot data
- Original hypothesis
- Experimental details and design of proposed investigation
- If requesting funding for a clinical study, include all information listed in the Clinical Study Guidelines (Interventional Study) or in the Clinical Study Guidelines (Observational Study). If you do not provide the information outlined in the guidelines, your application may be returned without formal consideration.
- Power calculations
- Expected value of results
- List of references relevant to the proposed project
- List of recent relevant publications from supervisor
- Appendices should be a maximum of 3 sides of A4 containing only tables or figures essential to the understanding of the application. Please include a Gantt chart for your project. Research papers whether published, in press or in preparation may not be attached (although hyperlinks can be provided).
- Statement detailing the scientific techniques in which the candidate will be trained
- Candidate’s training programme listing skills to be acquired during the grant
Sections 2-9 must not exceed 6 A4 sides. The PDF file size must not exceed 20MB.
Please ensure Arial font size 12 is used. If this font size is not used, the application will be rejected prior to formal consideration.
Additional document uploads that will be requested within pages on the application form:
- Full CV of the candidate including list of publications and academic email address
- Supporting letters from supervisors, confirming support for the candidate
- Supporting letters from collaborators
- Intellectual property agreements where relevant
- Valid copy of ethical approval if relevant
- Quotes obtained for any requested equipment with a value of £5,000 or more
The BHF is prepared to consider funding or part-funding Clinical Research Training Fellowships that include research experience with an industry partner (for example, experience of experimental medicine). Please contact our office for advice if you are considering applying for a co-funded Clinical Research Training Fellowship.
When to apply
The next deadline for applications is 1 July 2026, 17:00. This is the deadline that your application must be fully submitted to BHF after being approved by your Head of Department and Administrative Authority. Applications submitted after this date will not be considered until the following deadline. The following application deadline is 13 January 2027. BHF reserves the right to modify deadline dates.
Decision process
Receipt will be acknowledged within 24 hours and we will advise you when you can expect the result. A decision will be reached by the Clinical Fellowships Committee after it has considered external peer review reports.
The flowchart below shows the application journey
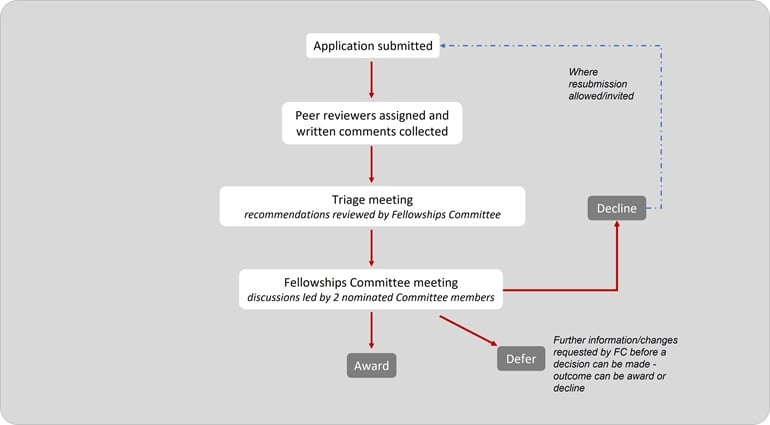
Independent expert review
To help applicants better understand how their proposals are assessed, please see BHF independent review guidelines and blank review form.
Apply for research grants

